Unyvero™ HPN (Hospitalised Pneumonia)
Rapid PCR Identification of Pathogens and Antimicrobial Resistance Genes
Turnaround Time: 6-8 hours
Brochure for Clinics: Download
Multiple Applications
Nosocomial and ventilator-associated pneumonia.
Multiple Sample Types
Sputum, bronchoalveolar lavage (BAL), tracheal aspirates.
Enables Expedient Clinical Actions
6-8 hours turnaround time. Supports antibiotic stewardship.
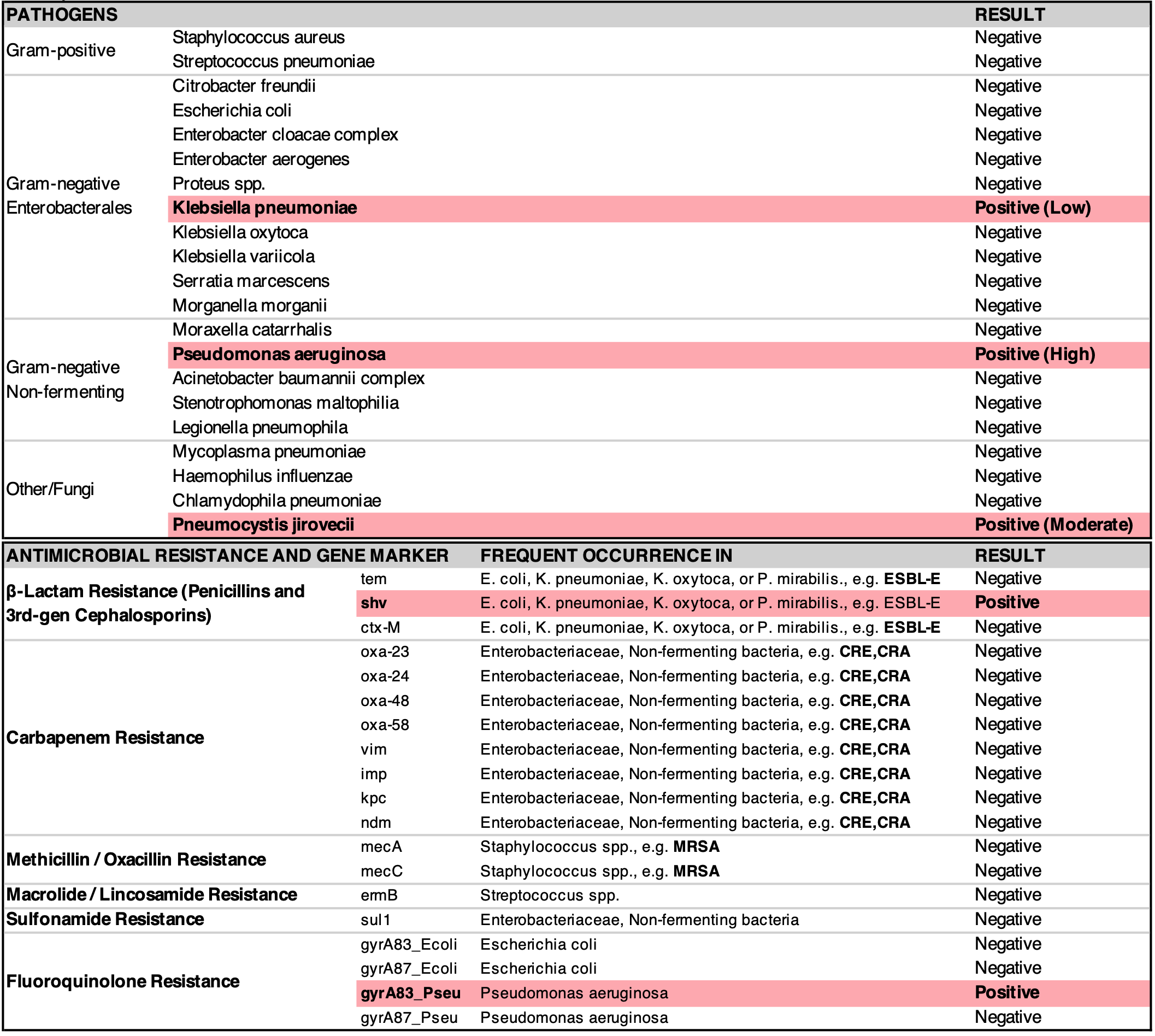
Result Report
In-a-glance reporting of all pathogens and antibiotic resistance genes.
Sample Report from a clinical test with sputum with reported lab turnaround time of 4 hr 40 min.
IDSA Therapeutic Guidelines Included as part of the Result Report
Disclaimers: The healthcare provider shall exercise professional judgement and careful interpretation of the test results and provided guidelines from local health regulatory bodies and the IDSA in determining their advice to the patient and in the dose selection of drugs.
Test Performance
2021, Karolinska, Sweden (Eur J Clin Microbiol Infect Dis.)
Overall Sensitivity: 95.1% (95%CI 96.5–98.3%)
Overall Specificity: 98.3% (95% CI 97.5–98.9%)
Overall PPV: 71.6% (95% CI 61.0–80.3%)
Overall NPV: 99.8% (95% CI 99.3–99.9%)
The Unyvero HPN panel was strongly concordant (98.2%) with bacterial culture for organism targets, confirming all pathogens reported by culture in 31 out of 35 patient samples. The Unyvero HPN panel further detected additional pathogens in 7 patients, one of which was captured by microbiologic culture 7 days later and confirmed positive, highlighting that the panel may detect pathogens missed by culture.
2020, North Carolina, USA (J Clin Microbiol.)
Overall PPA: 97.0%
Overall NPA: 99.9%
PPV for Antibiotic Resistance Markers: 100%
“…Positive PCR results from the LRT assay were largely concordant with culture results, suggesting that a rapid result could lead to appropriate or targeted antimicrobial therapy more quickly than quantitative culture. The most valuable utility of this assay may be for patients with HCAP/HAP/VAP or those who are critically ill or immunocompromised or have a disease that has been refractory to treatment.”
2021, 11 Clinical Trial Sites, USA (J Clin Microbiol.)
Overall PPA: 93.4%
Overall NPA: 98.3%
PPV for Antibiotic Resistance Markers: 80% - 100%
Performance evaluated using 1,016 prospectively collected and 392 archived specimens from 11 clinical trial sites in the United States. Additional potential pathogens were identified by Unyvero in 21.7% of prospectively collected specimens not detected in routine cultures.
2020, 3 ICUs, France (Critical Care)
Overall Sensitivity: 80% (95%CI 73–88%)
Overall NPA: 99% (95%CI 99–100%)
PPV for Antibiotic Resistance Markers: 100%
Performance evaluated using 95 clinical samples of VAP or ventilated HAP samples. Sensitivity was better for Gram-negative bacteria (90%) than for Gram-positive cocci (62%) (p = 0.005).
Among 17 empiric antibiotic treatments with carbapenems, 10 could have been de-escalated in the following hours according to Unyvero results.
Unyvero results also led to 2 unexpected diagnoses of severe legionellosis confirmed by culture methods.
Posted on March 27, 2020 by Curetis GmbH, Germany
Unyvero HPN Panel for Rapid Detection of Bacterial Co-Infections in Covid-19 Pneumonia
Bacterial co-infections in COVID-19 patients increase morbidity, mortality, and AMR threat.
Rapid diagnostic tests are critical in this public health response.
Bacterial co-infections are the hidden threat lurking behind COVID-19.
Early diagnosis of secondary bacterial infections limits adverse COVID-19 outcomes by informing timely and appropriate antibiotic treatment.
Rapid detection of bacterial infections protects high-risk patients and limits super-spreading in healthcare facilities.
Full release by manufacturer Curetis GmbH here.